Welcome to the
Abu-Remaileh Lab at Stanford University
We study the molecular and cellular bases of metabolic adaptation in health and disease
OUR RESEARCH
We are interested in identifying novel pathways that enable cellular and organismal adaptation to metabolic stress and changes in environmental conditions. We also study how these pathways go awry in human diseases such as cancer, neurodegeneration, and metabolic syndrome in order to engineer new therapeutic modalities.

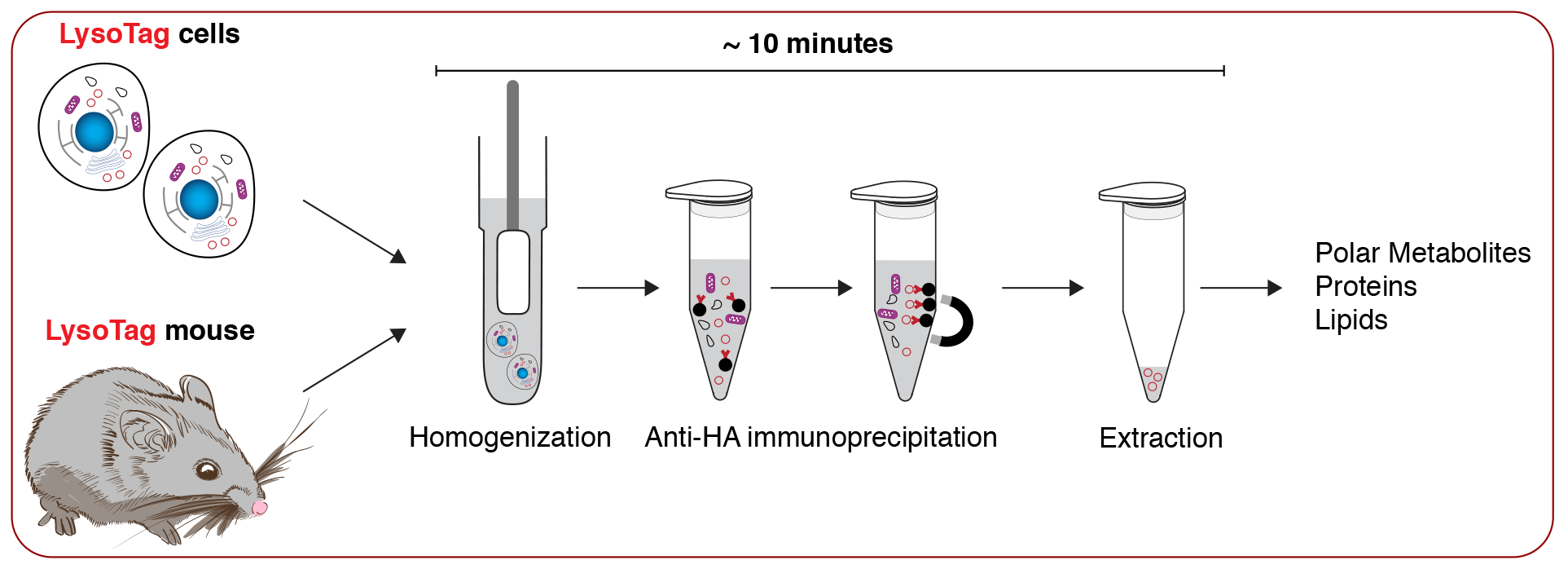
To address these questions, our lab uses a multidisciplinary approach to study the biochemical functions of the lysosome in vitro and in vivo. Lysosomes are membrane-bound compartments that degrade macromolecules and clear damaged organelles to enable cellular adaptation to various metabolic states. Lysosomal function is critical for organismal homeostasis—mutations in genes encoding lysosomal proteins cause severe human disorders known as lysosomal storage diseases, and lysosome dysfunction is implicated in age-associated diseases including cancer, neurodegeneration and metabolic syndrome.
By developing novel tools and harnessing the power of lipidomics, metabolomics, proteomics, and functional genomics our lab will define (1) how the lysosome communicates with other cellular compartments to fulfill the metabolic demands of the cell under various metabolic states and (2) and how its dysfunction leads to rare and common human diseases. Using insights from our research, we will engineer novel therapies to modulate the pathways that govern human disease.
PEOPLE

Monther Abu-Remaileh, Ph.D.
ChEM-H Institute Scholar
Assistant Professor of Chemical Engineering
Assistant Professor of Genetics
Email: monther (at) stanford.edu

Ali Goochani, Ph.D.
Basic Life Research Scientist
aligh (at) stanford.edu

Wentao Dong, Ph.D.
Basic Life Research Scientist
Email: wdong5 (at) stanford.edu

Jian Xiong, Ph.D.
Postdoctoral Scholar
xiongj (at) stanford.edu

Zhijun (Andrew) Zhu, Ph.D.
Postdoctoral Scholar
zjzhu (at) stanford.edu

Michael Fernandopulle, M.D., Ph.D.
Clinical Fellow
msfernan (at) stanford.edu

Cindy Lin
Ph.D. Student (Genetics)
clin5 (at) stanford.edu

Austin Murchison
Ph.D. Student (Biology)
austinmu (at) stanford.edu

George Walters-Marrah
Ph.D. Student (Biophysics)
gwmarrah (at) stanford.edu

Hisham Alsohybe
Ph.D. Student (Biophysics)
hishama (at) stanford.edu

Nick Manfred
Ph.D. Student (Neuroscience)
nickmanfred (at) stanford.edu

Shadler Nguyen
Ph.D. Student (Chemistry)
snguye11 (at) stanford.edu

Natalia Avina
Ph.D. Student (Cancer Biology)
navinaoc (at) stanford.edu

Matthew Matrongolo
Ph.D. Student (Neuroscience)
mjm4 (at) stanford.edu

Anastasiia Safronova
Ph.D. Student (Chemistry)
anasaf (at) stanford.edu

Chetan Chilkunda
Ph.D. Student (Chemical Engineering)
cchilkun (at) stanford.edu

Ibrahim Maaz
Ph.D. Student (Biochemistry)
imaaz (at) stanford.edu

Maryam Idris
Lab Manager
midris (at) stanford.edu
SELECTED PUBLICATIONS
For full list visit Google Scholar. ‡ denotes co-corresponding authors.
2026
Ghoochani A, Heiby JC, Rawat ES, Medoh UN, Di Fraia D, Dong W, Gastou M, Rastogi M, Hernandez V, Nyame K, Laqtom NN, Durso W, Valkova C, Isakova A, Kaether C, Wernig M, Gomez-Ospina N, Franke C, Ori A, Abu-Remaileh M. Cell-type resolved protein atlas of brain lysosomes identifies SLC45A1-associated disease as a lysosomal disorder. Cell. 2026 February 5; 189(3):765–782.e31.
2025
Nyame K, Xiong J, Alsohybe HN, De Jong APH, Peña IV, De Miguel R, Brummelkamp TR, Hartmann G, Nijman SMB, Raaben M, Simcox JA, Blomen VA, Abu-Remaileh M. PLA2G15 is a BMP hydrolase and its targeting ameliorates lysosomal disease. Nature. 2025 June 12; 642(8067):474–83.
Davidson JW, Jain R, Kizzar T, Geoghegan G, Nesbitt DJ, Cavanagh A, Abe A, Nyame K, Hunger A, Chao X, James I, Walesewicz H, Baldwin DA, Wade G, Michorowska S, Verma R, Schueler K, Hinkovska-Galcheva V, Shishkova E, Ding W-X, Coon JJ, Shayman JA, Abu-Remaileh M, Simcox JA. Hepatic lipid remodeling in cold exposure uncovers direct regulation of bis(monoacylglycero)phosphate lipids by phospholipase A2 group XV. Cell Metabolism. 2025 June 3; 37(6):1413-1425.e6.
Dong W, Nyame K, Rawat ES, Medoh UN, Xiong J, Bonin CC, Alsohybe HN, Liu HY, Gomes S, Hsieh T, Arnold M, Hsieh F, Sammler E, Abu-Remaileh M. Robust analytical methods for bis(monoacylglycero)phosphate profiling in health and disease. bioRxiv. 2025 Feb 17.
Saarela D, Lis P, Gomes S, Nirujogi RS, Dong W, Rawat E, Glendinning S, Zeneviciute K, Bagnoli E, Fasimoye R, Lin C, Nyame K, Boros FA, Zunke F, Lamoliatte F, Elshani S, Jaconelli M, Jans JJM, Huisman MA, Posern C, Westermann LM, Schulz A, Van Hasselt PM, Alessi DR‡, Abu-Remaileh M‡, Sammler EM‡. Tagless LysoIP for immunoaffinity enrichment of native lysosomes from clinical samples. Journal of Clinical Investigation. 2025 Feb 17; 135(4):e183592.
Kenny TC, Scharenberg S, Abu-Remaileh M‡, Birsoy K‡. Cellular and organismal function of choline metabolism. Nature Metabolism. 2025 Jan 8; 7(1):35–52.
2024
Medoh UN, Abu-Remaileh M. The Bis(monoacylglycero)-phosphate Hypothesis: From Lysosomal Function to Therapeutic Avenues. Annual Review of Biochemistry. 2024 Aug 2; 93(1):447–69.
Murchison AK, Abu-Remaileh M. Sharing is caring: TMEM165 a Golgi calcium importer used by the lysosome. Trends in Biochemical Sciences. 2024 Aug 1; 49(8):658–9.
Colella P, Sayana R, Suarez-Nieto MV, Sarno J, Nyame K, Xiong J, Pimentel Vera LN, Arozqueta Basurto J, Corbo M, Limaye A, Davis KL, Abu-Remaileh M, Gomez-Ospina N. CNS-wide repopulation by hematopoietic-derived microglia-like cells corrects progranulin deficiency in mice. Nature Communications. 2024 July 5; 15(1):5654.
Nyame K, Hims A, Aburous A, Laqtom NN, Dong W, Medoh UN, Heiby JC, Xiong J, Ori A, Abu-Remaileh, M. Glycerophosphodiesters inhibit lysosomal phospholipid catabolism in Batten disease. Molecular Cell. 2024 Apr 4; 84(7):1354-1364.e9.
Haney MS, Pálovics R, Munson CN, Long C, Johansson PK, Yip O, Dong W, Rawat E, West E, Schlachetzki JCM, Tsai A, Guldner IH, Lamichhane BS, Smith A, Schaum N, Calcuttawala K, Shin A, Wang Y-H, Wang C, Koutsodendris N, Serrano GE, Beach TG, Reiman EM, Glass CK, Abu-Remaileh M, Enejder A, Huang Y, Wyss-Coray T. APOE4/4 is linked to damaging lipid droplets in Alzheimer’s disease microglia. Nature. 2024 Apr 4; 628(8006):154–61.
2023
Medoh UN, Hims A, Chen JY, Ghoochani A, Nyame K, Dong W, Abu-Remaileh M. The Batten disease gene product CLN5 is the lysosomal bis(monoacylglycero)phosphate synthase. Science. 2023 Sept 15; 381(6663):1182–9.
Fasimoye R, Dong W, Nirujogi RS, Rawat ES, Iguchi M, Nyame K, Phung TK, Bagnoli E, Prescott AR, Alessi DR‡, Abu-Remaileh M‡. Golgi-IP, a tool for multimodal analysis of Golgi molecular content. PNAS USA. 2023 May 16; 120(20):e2219953120.
Scharenberg SG, Dong W, Ghoochani A, Nyame K, Levin-Konigsberg R, Krishnan AR, Rawat ES, Spees K, Bassik MC, Abu-Remaileh M. An SPNS1-dependent lysosomal lipid transport pathway that enables cell survival under choline limitation. Science Advances. 2023 Apr 21; 9(16):eadf8966.
Crossley MP, Song C, Bocek MJ, Choi JH, Kousouros JN, Sathirachinda A, Lin C, Brickner JR, Bai G, Lans H, Vermeulen W, Abu-Remaileh M, Cimprich KA. R-loop-derived cytoplasmic RNA–DNA hybrids activate an immune response. Nature. 2023 Jan 5; 613(7942):187–94.
2022
Armenta DA, Laqtom NN, Alchemy G, Dong W, Morrow D, Poltorack CD, Nathanson DA, Abu-Remalieh M‡, Dixon SJ‡. Ferroptosis inhibition by lysosome-dependent catabolism of extracellular protein. Cell Chemical Biology. 2022 Nov 17; 29(11):1588-1600.e7.
Laqtom NN, Dong W, Medoh UN, Cangelosi AL, Dharamdasani V, Chan SH, Kunchok T, Lewis CA, Heinze I, Tang R, Grimm C, Dang Do AN, Porter FD, Ori A, Sabatini DM, Abu-Remaileh M. CLN3 is required for the clearance of glycerophosphodiesters from lysosomes. Nature. 2022 Sept 29; 609(7929):1005–11.
Pedram K, Laqtom NN, Shon DJ, Di Spiezio A, Riley NM, Saftig P, Abu-Remaileh M, Bertozzi CR. Lysosomal cathepsin D mediates endogenous mucin glycodomain catabolism in mammals. PNAS USA. 2022 Sept 27; 119(39):e2117105119.
Dong W, Rawat ES, Stephanopoulos G‡, Abu-Remaileh M‡. Isotope tracing in health and disease. Current Opinion in Biotechnology. 2022 Aug; 76:102739.
Medoh UN, Chen JY, Abu-Remaileh M. Lessons from metabolic perturbations in lysosomal storage disorders for neurodegeneration. Current Opinion in Systems Biology. 2022 Mar; 29:100408.

JOIN US!
Graduate Students:
Current Stanford graduate students from all programs (PhD, MD, PhD/MD) who are interested in joining our lab are welcome to contact Monther directly. Please send a current CV in pdf format.
Postdocs and Clinical Fellows:
Motivated postdocs and clinical fellows with expertise in biochemistry, molecular biology, metabolism, mouse physiology or neurobiology are encouraged to apply. Please email a cover letter, your CV, and contact information for three references to Monther.
Undergradaute Students:
Students who are interested in conducting biomedical research and being part of our multidisciplinary team are welcome to email Monther for opportunities.
Master Students:
Current masters students who are enrolled in national or international programs and hoping to join our lab as visiting students (6-12 month duration) to complete their thesis are welcome. Research experience in biology, biochemistry or chemistry is an advantage. Please email a cover letter, your CV, and contacts of two references to Monther.
CONTACT US!
Address:
ChEM-H Building, Room: N227
290 Jane Stanford Way
Stanford, CA 94305

Our Lab is located at the new building that hosts both ChEM-H and Wu Tsai Neuroscience Research Center.